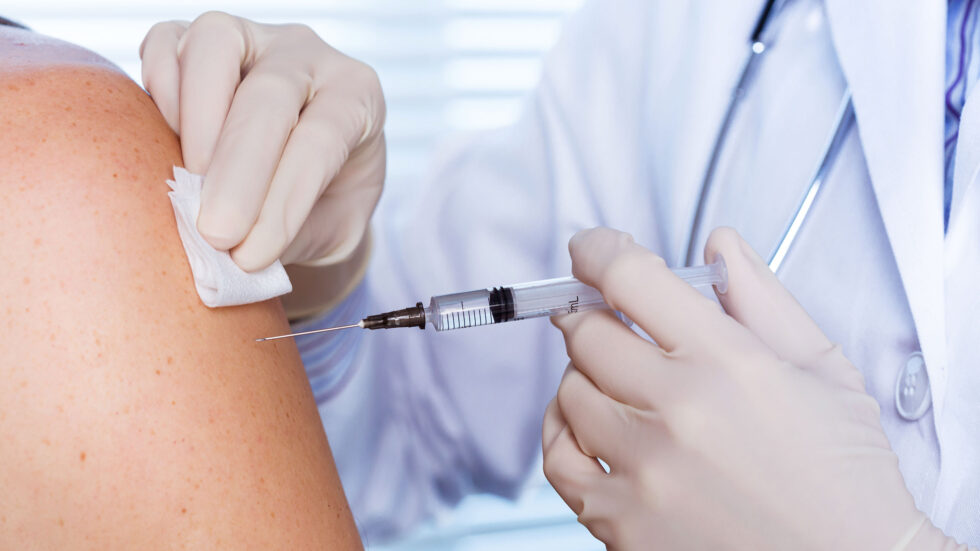
As researchers race to develop a COVID-19 vaccine, two Rutgers Global Health Institute core faculty members discuss how clinical trials work, the ethics of developing and distributing a vaccine, safety and efficacy in clinical trials, and what a successful vaccine may mean. Associate Professor Shobha Swaminathan is principal investigator at the Rutgers New Jersey Medical School Clinical Research Center and the medical director of Infectious Diseases Practice at University Hospital, and Michael K. Gusmano is a bioethicist and professor of health policy at the Rutgers School of Public Health. To read the full story.
Recent Posts
- NJACTS Community Engagement Core Available Services
- Unveiling the Biomechanical Forces that Drive Scarring.
- Scientists Discover Class of Crystals With Properties That May Prove Revolutionary.
- Will Banning Food Dyes Improve Our Health?
- Backed by Science and Evidence, New Jersey’s Actions Aim to Advance Maternal Health.
Categories
- Community (2,290)
- Covid (987)
- CTO Events (6)
- News (2,915)
- Pilots (21)